GENE THERAPY
Gene therapy is a groundbreaking approach to treating diseases by modifying patient’s genetic material, often using viral vectors like AAV or lentiviruses.
Gene editing to clinical validation, gene therapy products requires a robust and streamlined workflow. Allianz Bio, in partnership with ACRO, offers a state-of-the-art product line to support every stage of gene therapy development. From vector design to quality control.
Their portfolio includes high-quality reagents such as GMP-grade proteins, AAV Titer Quantitation kits, and GenPower™ enzymes for efficient vector production and gene editing.
To ensure product safety and regulatory compliance, ACRO provides resDetect™ kits for sensitive detection of residual host cell DNA, HCPs, and nucleic acids. Additionally, ClinMax™ ELISA kits enable accurate immune monitoring by measuring cytokines and biomarkers.
Together, these tools streamline gene therapy workflows, ensuring reliable, reproducible, and regulatory-ready results from research through preclinical development.
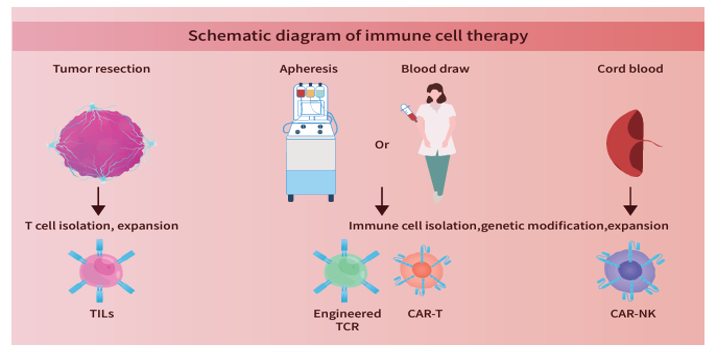
| T cell engineering | CAR evaluation | Kits |
|---|---|---|
| • Nucleic acid removal | • Car protein target | • Residual kit |
| • Gene editing enzyme (AAV titration kit) | • MHC molecules | • ELIspot kit |
| • Anti-CD19 CAR | • Biomarker ELISA kit | |
| • Car linker antibody | ||
| • Car linker antibody |
Key Feature
- End-to-End Workflow Support – Covers every stage from vector design to clinical validation in gene therapy development.
- High-Quality Reagents – Includes GMP-grade proteins, GenPower™ enzymes, and AAV Titer Quantitation kits for efficient vector production.
- Advanced QC Tools – resDetect™ kits ensure safety by detecting residual host cell DNA, HCPs, and nucleic acids.
- Immune Monitoring – ClinMax™ ELISA kits measure cytokines and biomarkers for accurate immune response analysis.
- Regulatory-Ready Solutions – Designed to deliver reliable, reproducible results aligned with preclinical and clinical standards.